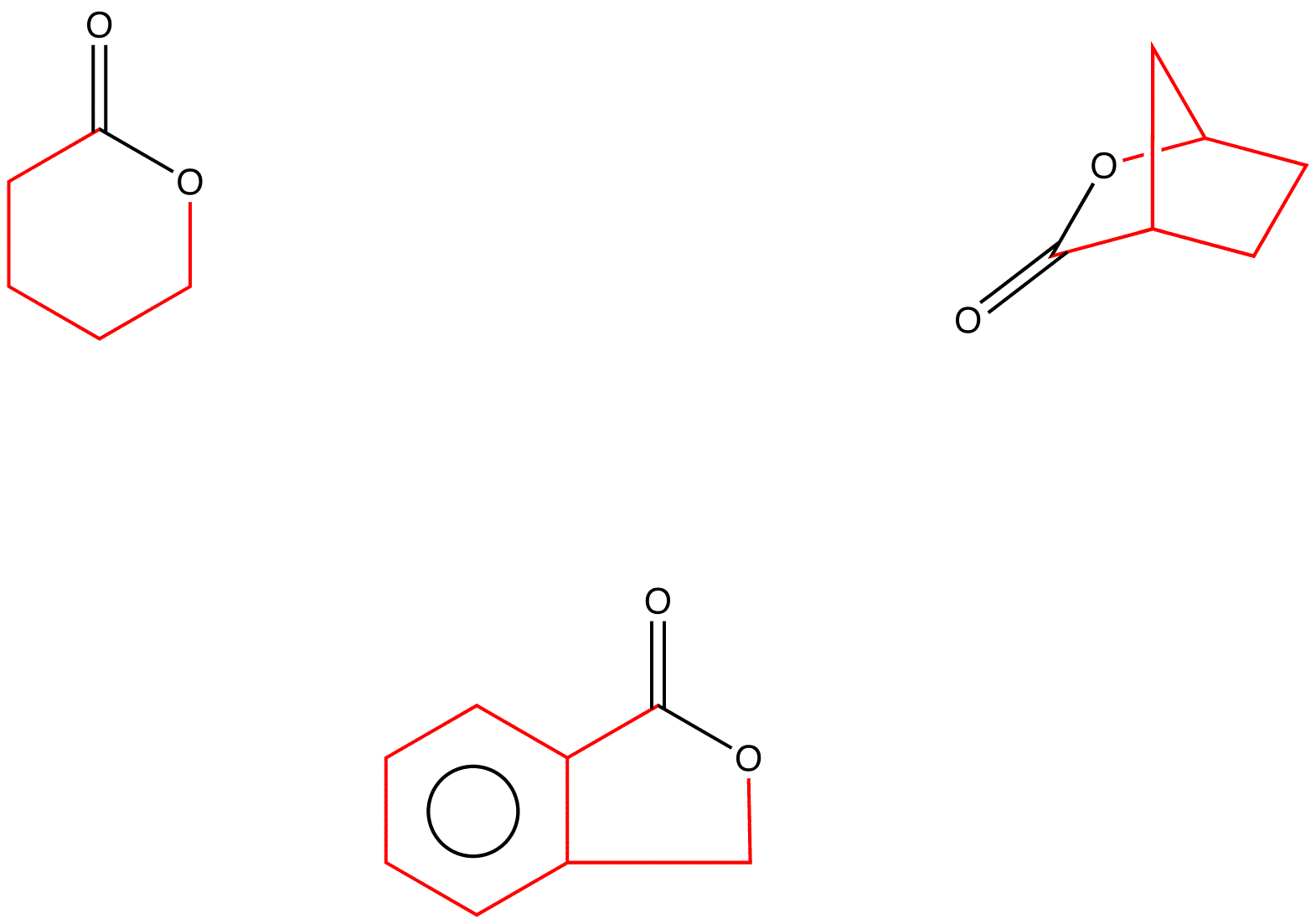
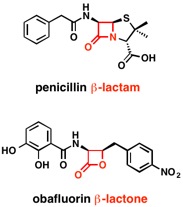
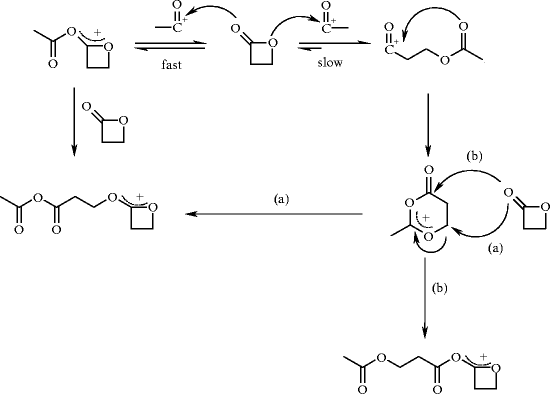
Organocatalysis applied to the ring‐opening polymerization of β‐lactones: A brief overview - Khalil - 2019 - Journal of Polymer Science Part A: Polymer Chemistry - Wiley Online Library

SciELO - Brasil - Synthesis of medium ring and macrocyclic acetylenic lactones by the ring expansion of oxabicycloalkenones Synthesis of medium ring and macrocyclic acetylenic lactones by the ring expansion of oxabicycloalkenones
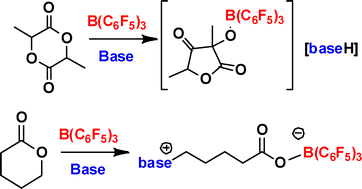
Ring openings of lactone and ring contractions of lactide by frustrated Lewis pairs - Dalton Transactions (RSC Publishing)

Mild and Chemoselective Lactone Ring-Opening with (TMS)ONa. Mechanistic Studies and Application to Sweroside Derivatives | The Journal of Organic Chemistry

Molecules | Free Full-Text | Lactones 41. Synthesis and Microbial Hydroxylation of Unsaturated Terpenoid Lactones with p-Menthane Ring Systems
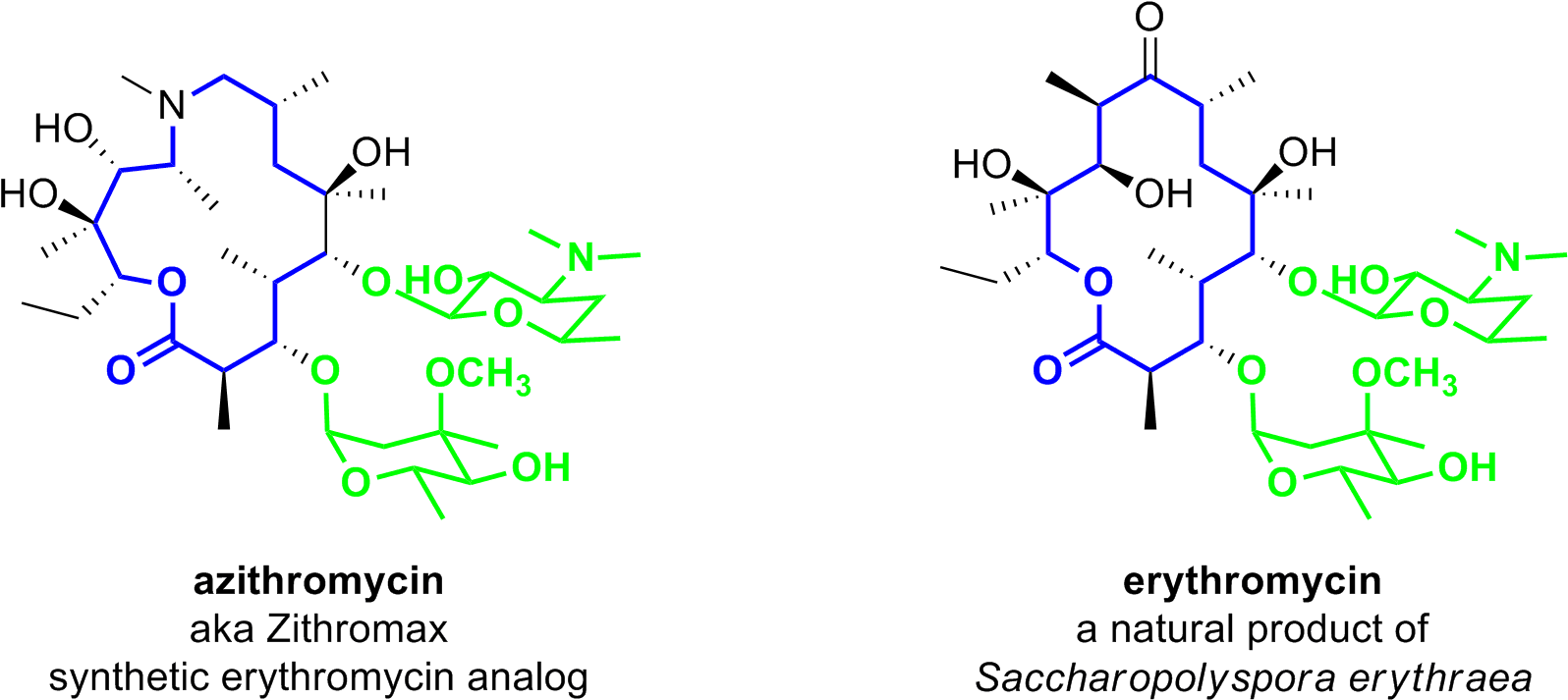
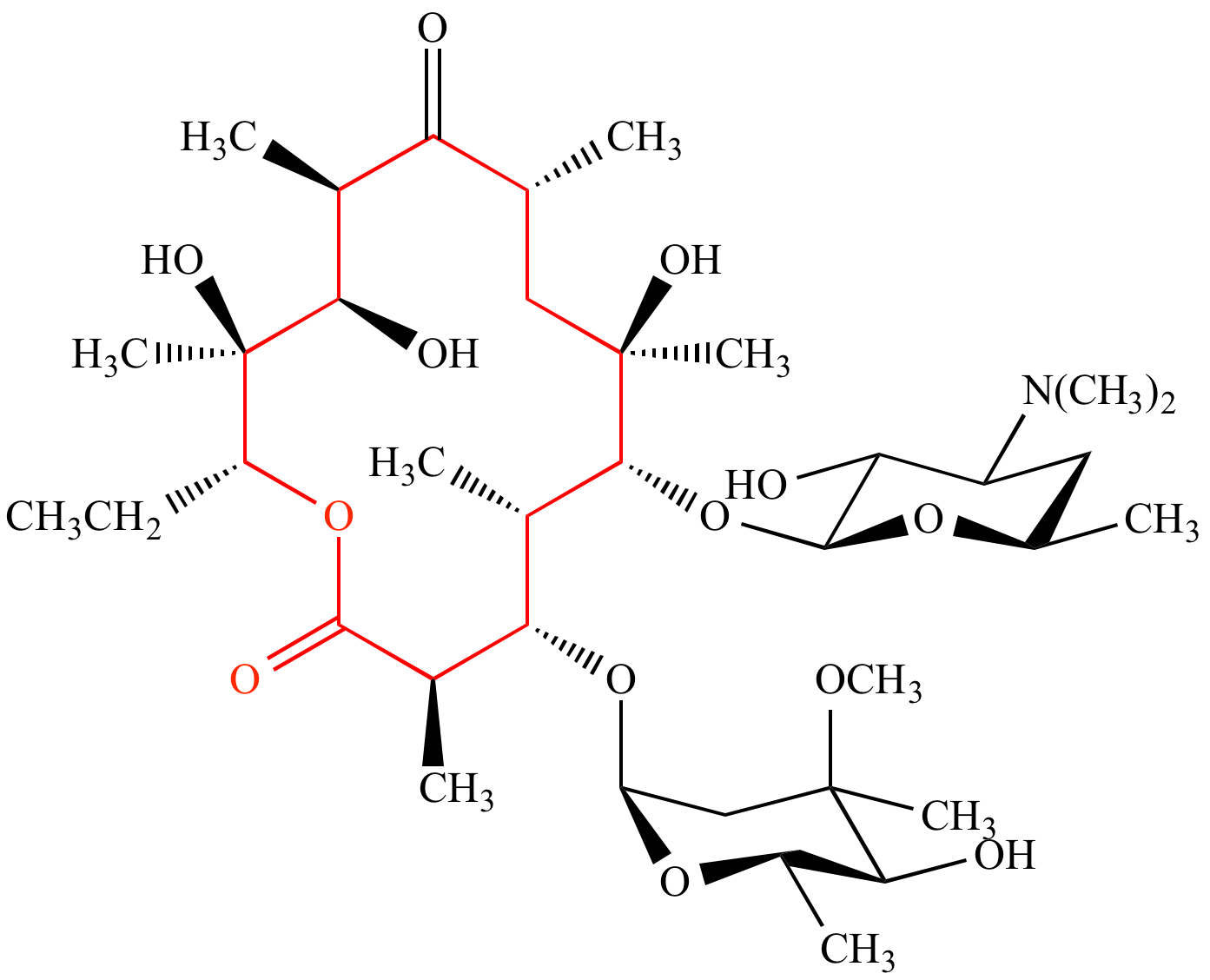
Download Macrolide-antibiotics - Lactone Ring In Azithromycin PNG Image with No Background - PNGkey.com

Size range of lactone rings produced by TE domains. The lactone ring of... | Download Scientific Diagram

Mild and Chemoselective Lactone Ring-Opening with (TMS)ONa. Mechanistic Studies and Application to Sweroside Derivatives | The Journal of Organic Chemistry
Structural Basis of the γ-Lactone-Ring Formation in Ascorbic Acid Biosynthesis by the Senescence Marker Protein-30/Gluconolactonase | PLOS ONE
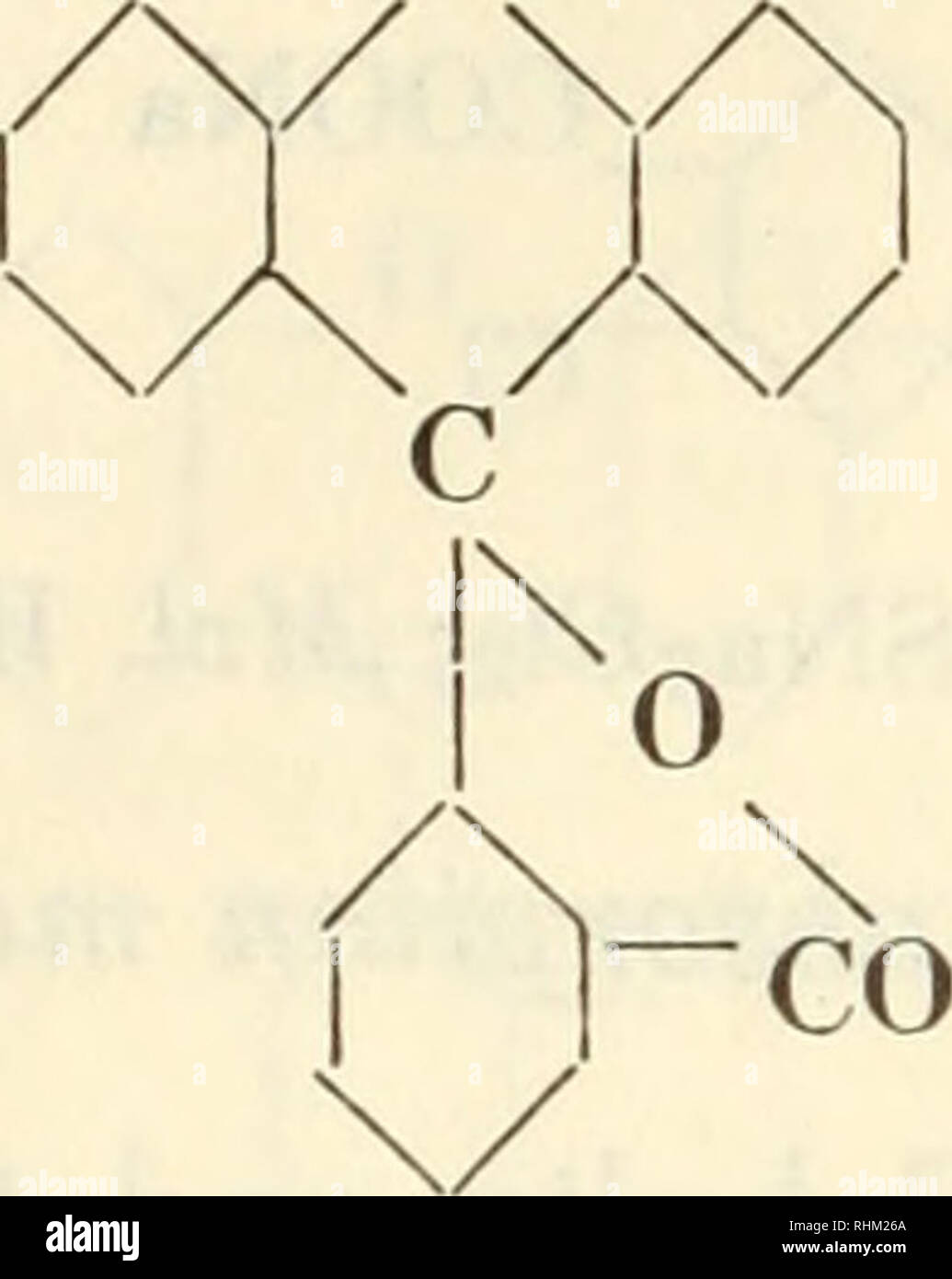
Biological stains; a handbook on the nature and uses of the dyes employed in the biological laboratory. Stains and staining (Microscopy); Stains and Staining; Dyes. 184 Biological Stains 3. FLUORAN DERIVATIVES

Lactones: Classification, synthesis, biological activities, and industrial applications - ScienceDirect

Organocatalysis applied to the ring‐opening polymerization of β‐lactones: A brief overview - Khalil - 2019 - Journal of Polymer Science Part A: Polymer Chemistry - Wiley Online Library